


Electroplate Heavy Metals Removal Wastewater Treatment Plant
Electroplating wastewater is not just a regulatory problem—it is a resource and a liability. the electroplate heavy metals removal treatment plant is no longer an optional expense—it is an essential pillar of sustainable manufacturing.
Electroplating wastewater always has heavy metal like Ni, Cu value over discharge standards
The classic treatment process of Electropate heavy metals wastewater as below:
Wastewater——Bar Screening------Dosing System for Ni remove/———Dosing System for Cu remove /——— Anaerobic Tank (MBBR)——Aerobic Tank (MBBR) ——MBR Tank ——Clean Water Tank ——Equipment Room-----sand/carbon----RO System
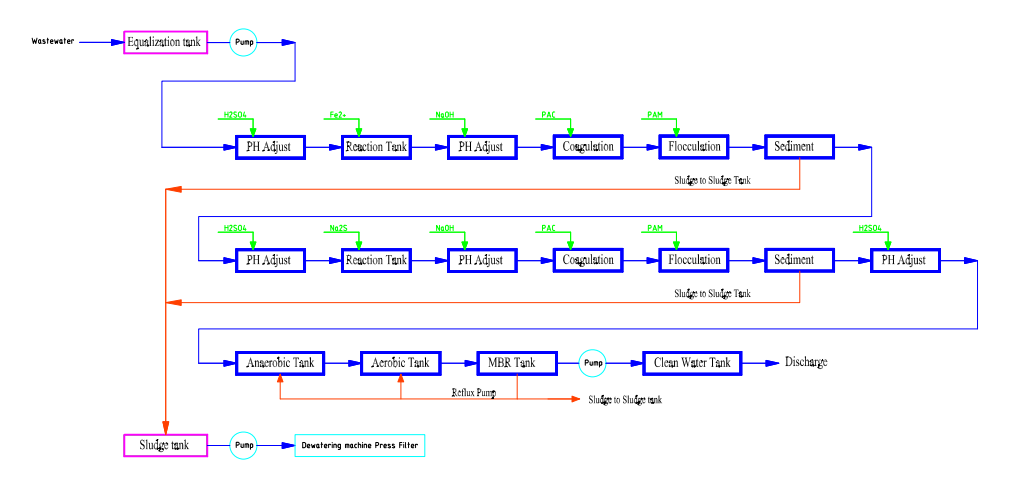
Dosing System (Ni Removing)
For electroplating nickel-containing wastewater and electroless nickel plating wastewater, chemical precipitation can be used for treatment. This method does not require complex equipment. Specifically, for electroplating nickel-containing wastewater, alkali can be directly added to adjust the pH to 11, followed by the addition of PAC (polyalanine chloride) and PAM (polyacrylamide) for coagulation and sedimentation. This process allows the nickel content to meet the discharge standards. If the nickel-containing wastewater is mixed with pretreatment wastewater, a small amount of heavy metal capture agent should be added after alkalization to facilitate chelation. The heavy metal capture agent can reduce nickel ion concentrations from low levels to compliant levels.
Reaction Principle:
Ni²⁺ + 2OH⁻ → Ni(OH)₂ (↓)
Reaction Condition: pH = 10–11
For nickel-containing wastewater with complexing agents, the first step should be to break down the complexes, followed by chemical precipitation.
Dosing System (Cu Removing)
copper is easily oxidized from the +1 valence state to the +2 valence state, although the stability constant of the Cu(CN)₃²⁻ complex ion is relatively large, divalent copper cannot form a stable complex with cyanide ions. Therefore, Cu(CN)₃²⁻ is still readily oxidized, resulting in the conversion of +1 valence copper to +2 valence copper and the oxidation of cyanide. In contrast, Fe(CN)₆⁴⁻ is different. Due to its relatively large stability constant, it is not easily oxidized under conditions of low effective chlorine concentration or low reaction temperature. When the reaction conditions are intensified to oxidize +2 valence iron to +3 valence, since Fe(CN)₆³⁻ remains very stable, the cyanide ions do not dissociate and are not oxidized.
Reaction Principle:
Cu²⁺ + 2OH⁻ → Cu(OH)₂ (↓)
Reaction Condition: pH = 9.5–10.5
For copper-containing wastewater with complexing agents, the first step should be to break down the complexes, followed by chemical precipitation.
Complex Breaking Reaction
• Sodium Sulfide (Na₂S)
The principle of the Na₂S method is as follows:
Na₂S + Cu[M] → CuS↓ + Na₂[M]
Sulfide (sodium sulfide) is added to wastewater containing complexed copper, followed by sodium hydroxide to adjust the wastewater pH to between 9 and 10. An appropriate amount of polyacrylamide (PAM) is then added to form copper sulfide (CuS), a precipitate with very low solubility. Under the action of PAM, copper ions are removed from the wastewater. The sulfide precipitation method can reduce the copper content in wastewater containing complexed copper to below 0.5 mg/L.
Further more details information about the treat process please contact with Hinada.